We recommend reading "How are Commercial Spirits Made?" prior to reading this article, as it provides a good overview of the topic of distillation. For those already familiar with the basics, read on!
The Boiling Temperature of Ethanol
We get a lot of questions about distillation and temperature. There is a lot of information stating that the boiling temperature of ethanol is 174 degrees Fahrenheit. However, this is only partially true. The boiling temperature of pure ethanol is 174 degrees F. The boiling temperature of ethanol in a wash, that is to say, ethanol mixed with water, is completely dependent on the ratio of ethanol to water. The more water there is in the solution, the higher the boiling temperature.
Commercial distillers that measure liquid temperature while distilling may have noticed that liquid temp can often be much higher than 174 before a still starts producing. Again, the boiling temperature of a solution that is 100% ethanol is 174 degrees. However, if a commercial distiller has a solution that is 50% ethanol and 50% water, the boiling temperature will be significantly higher. In fact, in a 50/50 solution of ethanol and water, the boiling (liquid) temperature of ethanol will be about 180 degrees.
Should a Still Start Producing Alcohol At 174 Degrees Fahrenheit?
One question we hear a lot is "Should I see alcohol coming out of my still once the temperature reaches 174 degrees F?" The answer to this question is "No, a commercial distiller should not." Why? As we mentioned, the boiling point of pure ethanol is 174 degrees F. Wash in a still is not pure ethanol. If it was, why would one be distilling it? A first run wash is generally never any stronger than 20% ethanol. The rest (80%) is water. More likely it's something like 10% alcohol (ethanol) and 90% water.
A solution that is 10% ethanol and 90% water is going to have a boiling temperature of about 197 degrees Fahrenheit. A wash that only has a starting alcohol of 10% ethanol won't boil anywhere near 174. The temp will have to be much higher than that before alcohol starts coming out of the still. For those that don't know how to determine the alcohol % of a wash, read our How to Use a Hydrometer article.
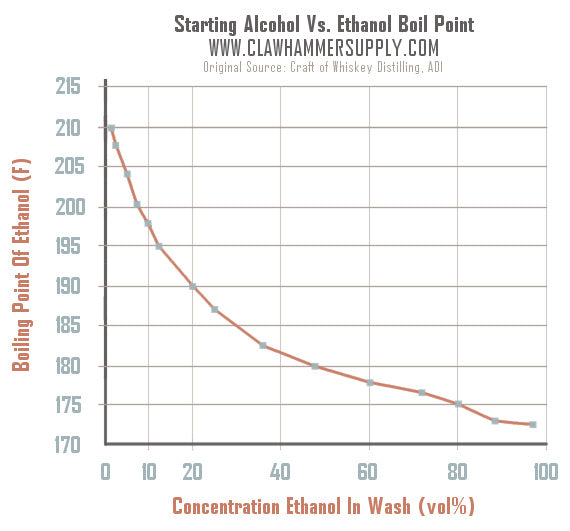
To determine the exact temperature at which ethanol will start boiling in a wash, check out this chart (Source: Craft of Whiskey Distilling by the American Distilling Institute). It shows the liquid boiling temp of ethanol based on the concentration of ethanol in a solution. For example, in a wash with a starting alcohol of 20%, ethanol won't begin to boil until liquid temp reaches 190 degrees F. Also, keep in mind, the data on the chart above only applies for distilling at sea level! The higher the elevation, the lower the boiling point.
Should a Still Maintain a Constant Temperature During Distillation?
Another question we hear is "should I keep my still temp exactly 174 F during a distillation run?" The answer to that is "no, absolutely not." Why? Well the answer has a lot to do with the chart above. For example, if a commercial distiller starts with a strong wash that has a starting alcohol of 20%, they can expect that ethanol will begin to boil out of the solution once liquid temp reaches about 190 F. As a commercial distiller let's their still run and more alcohol is extracted from the wash, the wash alcohol % drops accordingly. Lets say that half way through the run half of the alcohol has been taken out of the wash and the wash is at 10% abv. Well, at that point one can expect liquid temp to have increased to about 198 F. The bottom line is that as a still runs, the temp constantly increases.
Where Should a Thermometer Be Installed on a Still?
We prefer to install a temperature probe in the boiler, at the very least. Always make sure a 100% copper adapter and a stainless steel thermometer is being used. Adding a secondary thermometer at the top of the column to measure vapor temperature is helpful too. We recommend having one in both locations as it makes distilling a lot easier.


Thermometers at the top of the column
The two thermometers read different temperatures during the run, but they are also measuring two different things. The boiler temperature is measuring the temperature of the liquid inside the still, while the vapor thermometer is measuring the temperature of the vapor inside the column.
Keep in mind that the temperature reading is highly dependent on where the reading is being taken. First of all, never use an infrared thermometer to try and measure still temp. They aren't accurate. They measure surface temp of a still, not of the liquid or vapor inside. Also, if a still is polished and has a shiny surface, the signal could be bouncing off and giving a reading of ambient surfaces.
Should Vapor Temperature and Wash Temperature be the Same?
Vapor temp and wash temperature should be very different. Once vapor begins to form in the pot and it migrates up the column, the vapor temperature probe at the top of the column (if one is installed there) will rise from ambient temperature to 175 in a matter of a minute. Hypothetically speaking, at this point the boiler thermometer may read something like 195 F (again, depending on the starting alcohol) and the vapor probe could read as low as 175 F. There will always be a big difference between the two temperatures (boiler and vapor) which is not a big deal.
How to Use Temperature During Distilling
Temperature is mostly helpful determining when to seal the still, to know when it's about to start producing, and to know when it's about done producing. As far as making good product goes, we still think the most reliable method is to adjust heat based on the amount of product coming out of the still. A commercial distiller should be looking for steady, fast dripping, not a stream. Also, keep an eye on proof. If the proof is super low at the beginning of a run, there is either very low starting alcohol, or the still is running too hot. We use our parrot kit to constantly monitor the proof of the product coming out of the still.






Leave a comment